Structure of Atom Kit STEM Lab
Product Code : SCL-M-12461
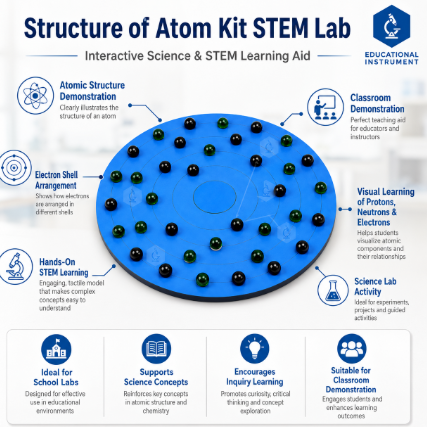
The Structure of Atom Kit STEM Lab is a premium educational aid designed to transform abstract chemistry theories into a tangible learning experience. Manufactured by Educational Instrument India, this kit provides a 3D visualization of atomic shells, sub-atomic particles, and chemical bonding. Based on Bohr’s Atomic Model, it allows students to physically arrange protons, neutrons, and electrons to understand atomic numbers, mass numbers, and the octet rule. It is an indispensable resource for chemistry laboratories, STEM centers, and secondary school classrooms.
Key Features & Educational Benefits:
3D Atomic Visualization: Provides a clear, physical representation of the nucleus and electron orbitals.
Hands-on Particle Physics: Includes color-coded beads or marbles representing Protons (Red), Neutrons (Yellow/Blue), and Electrons (Green) for easy identification.
Concept Mastery: Facilitates the teaching of complex topics such as valency, isotopes, ions, and the periodic table trends.
Curriculum Aligned: Perfectly suited for CBSE, ICSE, and international STEM frameworks (Grades 7–12).
Durable & Safe: Crafted from high-quality, non-toxic materials designed for years of classroom demonstration and student handling.
Product Specifications:
Brand: Educational Instrument India
Model Type: Bohr’s Atomic Model STEM Kit
Base Material: Reinforced polished wood or high-impact ABS plastic with defined orbital rings.
Components:
1 x Central Nucleus Base with Concentric Shells (K, L, M, N).
60+ Color-coded Sub-atomic Particles (Protons, Neutrons, Electrons).
Valency & Element Reference Cards.
Comprehensive Instructional Manual with element-building activities.
Dimensions: 30cm x 30cm (approximate lab standard).
Target Grade: Secondary and Higher Secondary (Ages 12+).
How to Use:
Define the Element: Select an element from the periodic table (e.g., Carbon).
Build the Nucleus: Place the correct number of Protons and Neutrons in the central well of the model.
Arrange Electrons: Following the 2n^2 rule, place the Electrons in the concentric shells (2 in the K-shell, 4 in the L-shell for Carbon).
Analyze Valency: Observe the outermost shell to determine the element's valency and its potential for chemical bonding.
Interactive Quiz: Use the included cards to challenge students to identify "Mystery Atoms" based on their particle arrangement.


.png)
.png)

.png)

.png)
